Introduction
The addendum to ICH E11 entitled, Clinical investigation on medicinal products in the pediatric population (E11), issued as Step 4 in 2017, highlights the recent advances in scientific and technical knowledge in pediatric medicinal product development. To further disseminate information on pediatric drug development (as part of medicinal product development), the Food and Drug Administration of the United States (U.S. FDA) and the Pharmaceuticals and Medical Devices Agency (PMDA) of Japan are pleased to announce the joint “PMDA-ATC & U.S. FDA Pediatric Review Seminar 2019” at the PMDA Office in Tokyo on July 8-11, 2019. This seminar is intended for regulatory authority officials who are engaged in the review of pediatric drug development programs.
The seminar will cover current pediatric guidelines and practices in the United States and Japan, and will provide the opportunity for the participants to share current pediatric guidelines and practices in their respective countries and regions. Case study sessions on pediatric drug development programs are also planned for small group discussions among the participants.
The aim of the seminar is to provide opportunities for the participants to review their own programs and to gather additional information that may enhance the review of pediatric drug development programs within their own regulatory frameworks.
The seminar is not open to the industry or non-regulatory authorities. Participation from many regulatory authorities is welcomed.
The training will be provided in English (with consecutive translation in some sessions). All participants are expected to actively participate in all the sessions.
Key Seminar Objective
By participating in this seminar, the participants will understand:
- Scientific, ethical and regulatory considerations when evaluating drug products being developed for the pediatric population
- Global standard guidelines relating to the review of drug products being developed for the pediatric population, e.g., ICH guidelines
- Current practice and issues in the review of drug products being developed for the pediatric population when data are collected in adults and the pediatric population of other races.
Who should apply
- Employee of regulatory authorities
- Pharmaceutical reviewers with experience in review of new drug applications (NDA) for pediatric population for over 3 years.
Date
July 8 to 11, 2019
Program (subject to change)
See the draft program (attached)
[Optional]
Participating regulatory authorities may request individual meetings with PMDA and FDA. These meetings will allow participating agencies to ask specific questions related to their agencies. Each meeting will be 30 minutes.
Registration (Registration is closed)
Number of participants: approx. 30
Close of registration: April 12, 2019.
- Registration request should be made by filling in all the necessary items including signature by Head of Organization on the application form (attached). Please send the form in both the Word file (without signature) and the PDF file (with signature) by e-mail to the e-mail address shown in “Contact Us” below.
- Early registration is recommended. After the close of registration, we are not able to accept change or addition of participant(s) in principle. Please be sure to allow enough time to arrange for the trip and obtain visa for entry to Japan.
- If the number of applications exceeds the capacity, the number of participants from each country may be limited. Selection of participants may take place according to their current work, work experience and expectations for this training course described in the application form.
- Confirmation of the registration and additional information will be sent to the approved participants after the close of registration.
- Please indicate if your agency would like to have an individual meeting with PMDA and FDA.
Fee
There is no registration fee for this seminar.
Information on travel and hotel reservation assistance will be announced to the approved participants.
Location
Pharmaceuticals and Medical Devices Agency (PMDA):
Shin-kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan
Access
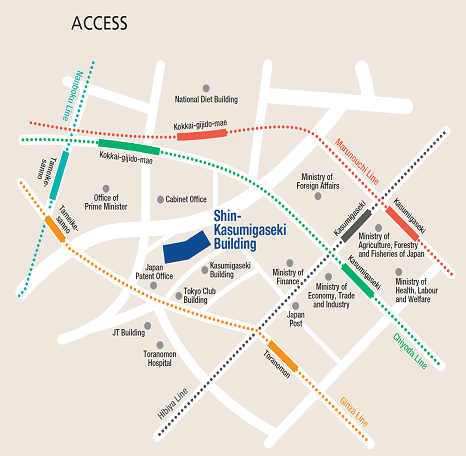
Please use public transportation. Nearest Tokyo Metro Subway Stations:
- 5 minutes' walk from Exit 11 of Toranomon Station on the Ginza Line
- 8 minutes' walk from Exit 3 of Kokkai-gijido-mae Station, on the Marunouchi Line, Chiyoda Line
- 8 minutes' walk from Exit A13 of Kasumigaseki Station, on the Marunouchi Line, Chiyoda Line, Hibiya Line
- 10 minutes' walk from Exit 8 of Tameike-sanno Station, on the Ginza Line, Namboku Line
Contact Us
For more information, please contact:
Secretariat, PMDA Asia Training Center
E-mail: PMDA-ATC●pmda.go.jp
Office of International Cooperation
Pharmaceuticals and Medical Devices Agency
Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan
(Note: For the purpose of security, @ in the e-mail addresses are replaced with ●. Please replace ● with @ when you send an e-mail.)
