Introduction
The Pharmaceuticals and Medical Devices Agency (PMDA) is pleased to announce the holding of the "PMDA-ATC Multi-Regional Clinical Trial (MRCT) Seminar 2020" for new drug application reviewers from overseas regulatory authorities. The seminar will be offered by the Asia Training Center for Pharmaceuticals and Medical Devices Regulatory Affairs (PMDA-ATC) at PMDA Office in Tokyo, from January 20-23, 2020. This seminar is held as a workshop of the Asia-Pacific Economic Cooperation, Life Sciences Innovation Forum, Regulatory Harmonization Steering Committee (APEC-LSIF-RHSC) Centers of Excellence; however, it is open to non-APEC economies as well.
With the increase in global product development, the ICH E17 guideline (General principle for planning and design of Multi-Regional Clinical Trials) has been finalized and its training material has been developed. Product reviews based on multi-regional clinical trials driven data are also increasing. Under this situation, it is essential that regulators adequately evaluate the benefit-risk balance while keeping in mind the ethnic factors.
The seminar aims to promote drug development and convergence of regulations by sharing accumulated experiences of Japan, such as protocol designing and planning of MRCT, clinical data evaluation, clinical operation, regulatory review based on results of GCP inspections, post-market safety evaluation of approved drugs based on MRCT. The seminar will be led not only by PMDA reviewers but also by representatives from academia and industry, allowing participants to think and discuss from multiple perspectives.
The seminar is not open to the industry or non-regulatory authorities. Participation from many regulatory authorities is welcomed.

Key Seminar Objectives
- To learn the current situation surrounding MRCTs
- To learn the key regulatory flow of the drug development to post-approval utilizing MRCTs
- To obtain an opportunity to discuss how to make regulatory decisions based on specific cases
- To build a skilled human capacity in regulatory science to promote and facilitate MRCTs
- To enhance the regulatory cooperation in the APEC region on the evaluation and regulation of MRCTs
- To increase the acceptability of MRCT data by multiple regulatory authorities
Who should apply
- Senior clinical reviewers with at least 3 years of hands-on experience in the assessment of clinical trial applications, benefit-risk profiles for market approvals and post-market activities
- The training will be provided in English (with consecutive translation in some sessions).
- All participants are expected to be actively involved in all of the sessions.
- A participant from each economy should present review systems of pharmaceuticals in his/her economy to other participants.
Date / Time
January 20 (Mon.) to 23 (Thu.), 2020
Program (subject to change)
See attached for the draft program
Registration (Registration is closed)
Number of participants: approx. 30
Close of registration: November 1 (Fri), 2019
Sending address : atc_seminar●nta.co.jp
(Note: For the purpose of security, @ in the e-mail addresses are replaced with ●. Please replace ● with @ when you send an e-mail.)
- Registration request should be made by filling in all the necessary items including signature by Head of Organization on the application form (attached). Please send the form in both the Word file (without signature) and the PDF file (with signature) by e-mail to the sending e-mail address above.
- Early registration is recommended. After the close of registration, we are not able to accept change or addition of participant(s) in principle. Also, please be sure to allow enough time to obtain the visa for entry to Japan.
- If the number of applications exceeds the capacity, the number of participants from each economy may be limited. Selection of the participants will take place at the discretion of PMDA based on the information provided in the application form.
- Confirmation of the registration and additional information will be sent to the approved participants after the close of registration.
Fee
There is no registration fee for this Seminar.
Information on travel and hotel reservation assistance will be announced to the approved participants.
Location
Pharmaceuticals and Medical Devices Agency (PMDA):
Shin-kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku,Tokyo 100-0013, Japan
Access
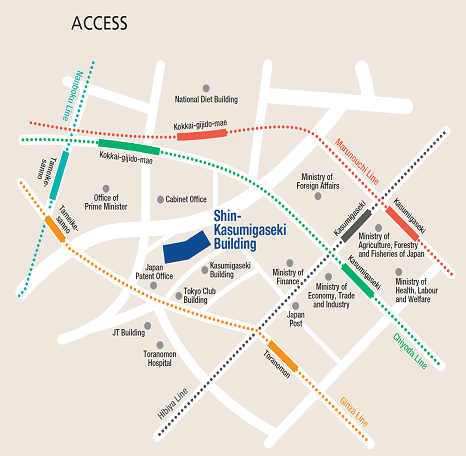
Please use public transportation. Nearest Tokyo Metro Subway Stations:
- 5 minutes’ walk from Exit 11 of Toranomon Station on the Ginza Line
- 8 minutes’ walk from Exit 3 of Kokkai-gijido-mae Station, on the Marunouchi Line, Chiyoda Line
- 8 minutes’ walk from Exit A13 of Kasumigaseki Station, on the Marunouchi Line, Chiyoda Line, Hibiya Line
- 10 minutes’ walk from Exit 8 of Tameike-sanno Station, on the Ginza Line, Nanboku Line
Contact Us
For more information, please contact:
Registration and Travel
Subcontractor, Nippon Travel Agency
E-mail: atc_seminar●nta.co.jp
Training contents
Secretariat, PMDA Asia Training Center
E-mail: PMDA-ATC●pmda.go.jp
Office of International Cooperation, Pharmaceuticals and Medical Devices Agency
Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan
(Note: For the purpose of security, @ in the e-mail addresses are replaced with ●. Please replace ● with @ when you send an e-mail.)
