The Drug Master File (DMF) System, allows the manufacturers of Active Pharmaceutical Ingredients (APIs) to submit the detailed information (manufacturing methods, data, etc.) of APIs to the Review Authority (PMDA).The registered information (manufacturing methods, data, etc.) is quoted as the necessary information for an approval review of the pharmaceutical products in which APIs is used.
DMF is reviewed at the time of the approval review for the pharmaceutical products quoting DMF. At the time of DMF registration, PMDA checks whether it is written in the correct format, e.g., minimum required items are included (application) and data is attached (CTD M3).
Notes) In Japan, the Drug Master File (DMF) is called “Master File” or “MF”.
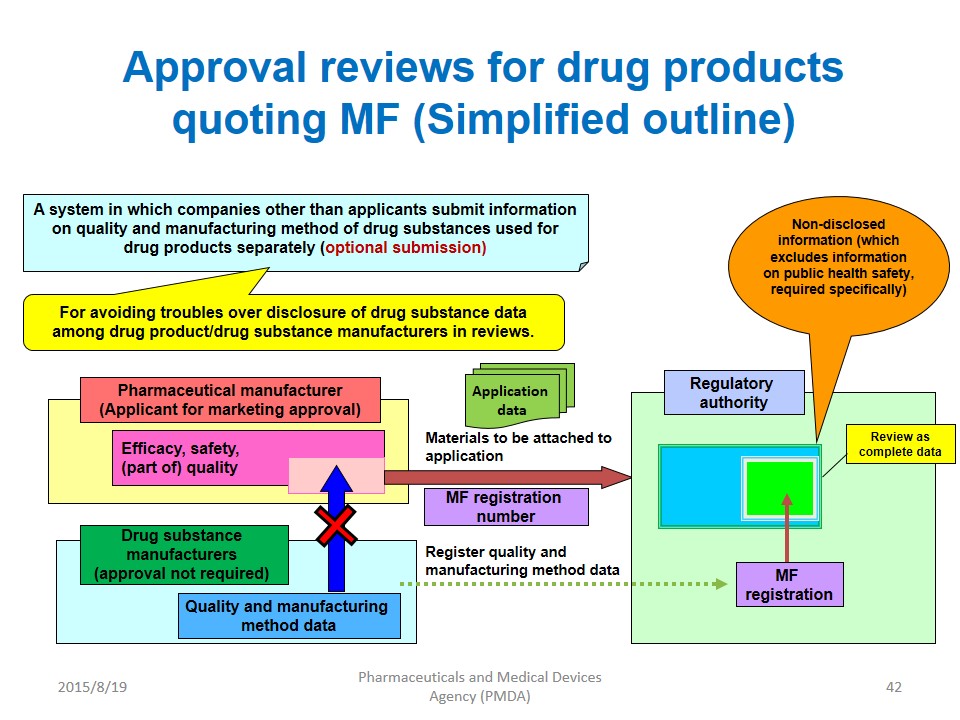
The Purpose of DMF is to protect the “know-how” of API manufacturing methods against the marketing authorization applicant (MAA) / holder (MAH) of pharmaceutical products. DMF is not a patent. The foreign manufacturers of APIs can apply for DMF registration. But the foreign manufacturers cannot apply directly to PMDA. To appoint “in-country caretakers of DMF” in Japan is Essential in PMDact. It is important for the DMF Holder, the in-country caretaker and related persons to understand the Japanese regulation and the guidance.
PMDact … the Act on Securing Quality, Efficacy and Safety of Pharmaceuticals, Medical Devices, Regenerative and Cellular Therapy Products, Gene Therapy Products, and Cosmetics
When we consider the review process for approval application of pharmaceutical products quoting DMF, the MAA/MAH, the DMF Holder and the in-country caretaker need to communicate with each other. In-country caretakers of DMF, who play important roles in the related administration procedures, etc. in Japan. Under the cooperation of the parties, PMDA review the submitted product applications, giving full consideration to the target review time.The target review time means the total review time or the regulatory review time. PMDA posts on its website the latest Annual Report, which discloses the Agency’s operating performance(The target review time).
- Master File System for Drug Substances, etc.
- Guidance on Drug Master File System in Japan
- Q&A on the Master File (MF) System, Part I
- Q&A on the Master File (MF) System, Part II
- Q&A on the Master File (MF) System, Part III
- Guideline on Utilization of Master File for Drug Substances, etc. PFSB / ELD Notification No. 0210004 February 10, 2005
Please "contact us" and make inquiries using the help request form if you find anything unclear in the above.
Please refer to Frequently Asked Questions (FAQ) as needed.
