Introduction
The Pharmaceuticals and Medical Devices Agency (PMDA) is pleased to announce the holding of the "PMDA-ATC Medical Devices Seminar 2016" for officials from overseas regulatory authorities who are engaged in the review of medical devices and in vitro diagnostics.
The Seminar will be offered by the Asia Training Center for Pharmaceuticals and Medical Devices Regulatory Affairs (PMDA-ATC) at PMDA Office in Tokyo, from November 7 to 11, 2016.
The Seminar will cover wide range of topics, such as regulations, consultations (scientific advices), product reviews, GCP/GLP/QMS, safety measures, standards, etc. Small group discussions among participants, and site visit to manufacturing facilities are planned as well.
The aims of the Seminar are to provide chances for the participants to refer back to their work and to find any additional opportunity for enhancement of the participant's regulatory system.
The seminar is not open to the industry or non-regulatory authorities. The seminar is not intended for Japanese government employees, either.
Participation from many regulatory authorities is welcomed.
Key Seminar Objectives
- To learn basics of medical device regulations and regulatory organization
- To learn the key regulatory flow of the review of medical devices, especially on the following
points.
- Product classifications based on risk
- Scientific reviews
- GCP/GLP/QMS/standards
- Safety measures
- To have discussions among participants using cases to experience the details of the learnings
from lectures
- To obtain updated information about international medical device activities (utlization of
international standards, IMDRF, etc)
- To observe the efforts at the manufacturing site to comply with the regulation
- To make use of the learnings / findings from the Seminar for the betterment of the regulatory
system in the participant’s organization.
Who should apply
- Officials from regulatory authorities who are currently engaged in review of medical devices and
in-vitro diagnostics.
A basic knowledge of the regulations pertaining to the medical devices product review
in his/her organization is required.
The training will be provided in English (with simultaneous translation in some sessions).
All participants are expected to actively participate in all of the sessions.
Date / Time
November 7 to 11, 2016.
Program (subject to change)
See attached for the draft program
Registration *Registration has closed*
- Registration request should be made by filling in all the necessary items on the application form
- Registration will close on September 30, 2016.
- We recommend you to register early. We may need to close the registration before the deadline
- Please be sure to allow enough time to obtain your visa.
- If the number of applications exceeds our capacity, we may limit the number of participants
- Confirmation of the registration and additional meeting information will be sent to the approved
Fee
There is no registration fee for this seminar.
Information on travel and hotel reservation assistance will be announced to the approved participants.
Location
Pharmaceuticals and Medical Devices Agency (PMDA):
Shin-kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku,Tokyo 100-0013, Japan
Access
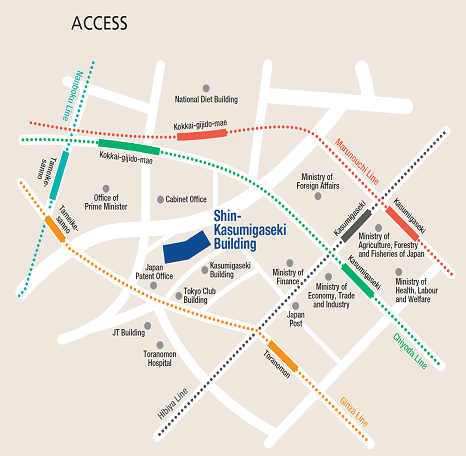
Please use public transportation. Nearest Tokyo Metro Subway Stations:
- 5 minutes’ walk from Exit 11 of Toranomon Station on the Ginza Line
- 8 minutes’ walk from Exit 3 of Kokkai-gijido-mae Station, on the Marunouchi Line, Chiyoda Line
- 8 minutes’ walk from Exit A13 of Kasumigaseki Station, on the Marunouchi Line, Chiyoda Line, Hibiya Line
- 10 minutes’ walk from Exit 8 of Tameike-sanno Station, on the Ginza Line, Nanboku Line
Contact Us
For more information, please contact:
Secretariat, PMDA Asia Training Center
E-mail: PMDA-ATC@pmda.go.jp
Office of International Cooperation
Pharmaceuticals and Medical Devices Agency
Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan
