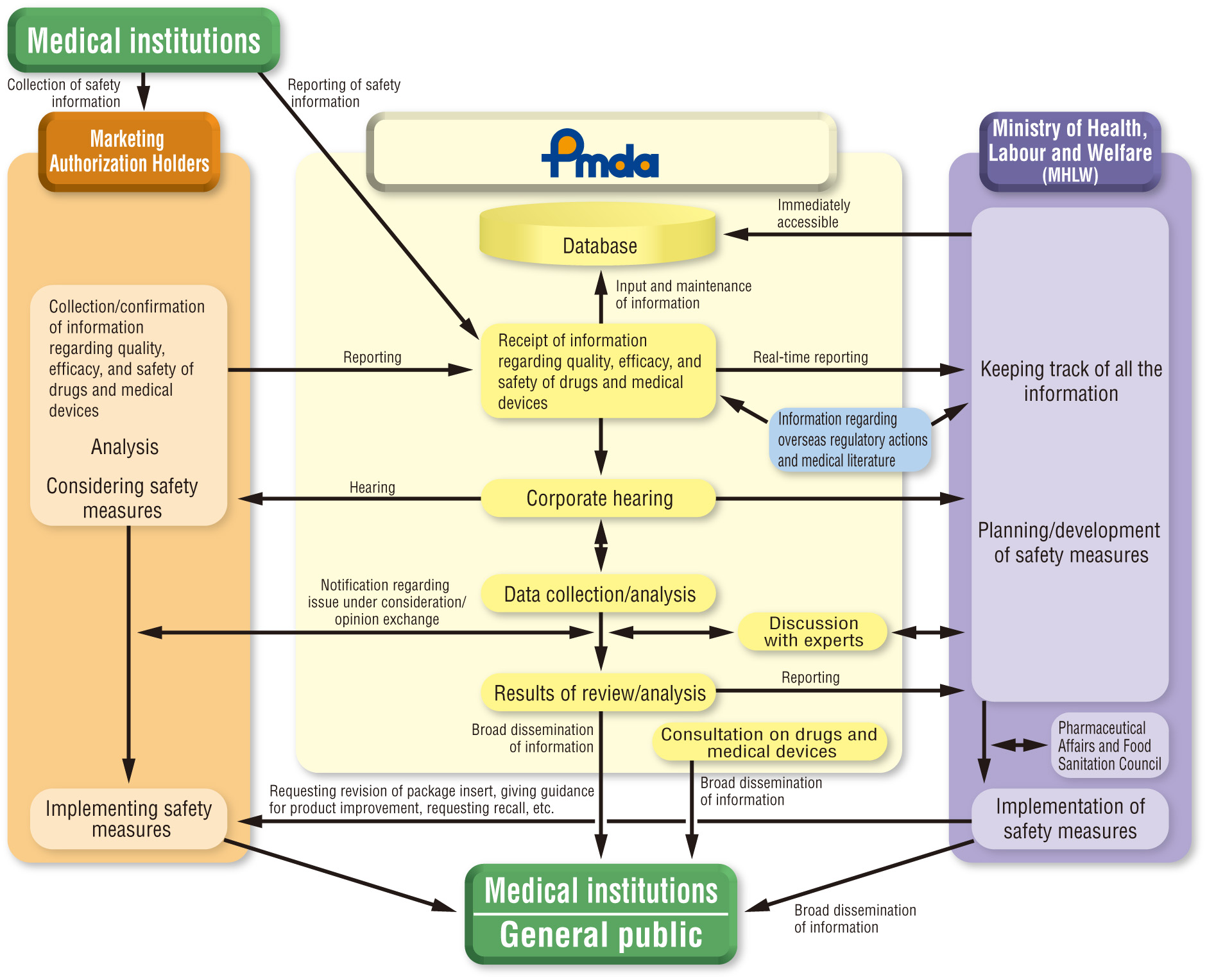
- We collect safety information, such as adverse drug reactions, infections caused by use of pharmaceuticals and medical devices and adverse events caused by medical devices from companies and healthcare professionals. That information is stored into a database for scientific analysis and investigation. Findings from investigation are reported to the MHLW, which would result in administrative actions to ensure safety use of pharmaceuticals and medical devices, etc.
- We develop and implement new methods and techniques for post-marketing safety operations, such as data mining method and sentinel medical institution network, with a view to enhancing quick response to problems and taking accurate safety measures. Thus it enables us to prevent and predict safety problems.
- We provide information on pharmaceuticals and medical devices on the website in order to promote their proper use, by listing package inserts, product recalls, drug guide for patients and other urgent safety alerts as well as reports on adverse drug reactions and adverse events of medical devices submitted to the PMDA from companies. We also offer free e-mail information delivery services providing latest safety information to the health care professionals.
- We provide telephone consultation services on pharmaceuticals and medical devices for consumers, etc.
- We collect safety measure contributions from marketing authorization holders of pharmaceuticals and medical devices.
- Home
- Post-marketing Safety Measures
- Outline
Post-marketing Safety Measures