Introduction
The Pharmaceuticals and Medical Devices Agency (PMDA) is pleased to announce the holding of the “PMDA-ATC Pharmacovigilance Seminar 2019” for overseas regulatory authority officials who are engaged in pharmacovigilance activities. The seminar will be offered by the Asia Training Center for Pharmaceuticals and Medical Devices Regulatory Affairs (PMDA-ATC) at the PMDA Office in Tokyo, on February 4 - 7, 2019.
The Seminar is held as a workshop of Asia-Pacific Economic Cooperation, Life Sciences Innovation Forum, Regulatory Harmonization Steering Committee (APEC-LSIF-RHSC) Center of Excellence; however, the Seminar is open to non-APEC economies as well.
The Seminar will cover wide range of topics on pharmacovigilance, including internationally recognized regulations, global safety data collection and evaluation of accumulated safety data, risk management based on the risk management plan including pharmacovigilance plan and risk minimization action plan including risk communication like labeling per safety specification. Group discussions among participants using case studies are planned as well.
The aims of the Seminar are to provide chances for the participants to refer back to their work and to find any additional opportunity for enhancement of the regulatory system in participants’ economies for pharmacovigilance in the post-marketing phase.
The Seminar will be led not only by PMDA staff members from pharmacovigilance area but also by representatives from U.S.FDA, academia and the pharmaceutical industry, allowing participants to think and discuss from multiple perspectives on pharmacovigilance.
The seminar is not open to the industry or non-regulatory authorities.
Participation from many regulatory authorities is welcomed.

Key Seminar Objectives
By participating in this workshop based on the core curriculum endorsed by APEC-LSIF-RHSC, the participants will learn the following knowledge that they could utilize in order to enhance the pharmacovigilance system in their countries/regions:
- The importance of harmonized regulatory strategy in ICH and CIOMS and regulatory updates to ensure compliance with new pharmacovigilance, risk management, and adverse event reporting, etc.,
- The risk management tools for pharmacovigilance and risk minimization action per safety specifications identified by signal detection and benefit-risk analysis,
- The importance of collection and accumulation of adverse drug reaction reports and methodology of signal detection using such accumulated data, from the point of view of pharmacoepidemiology, and
- The Benefit-Risk analysis through the life cycle of marketed medicinal products and the appropriate system for providing the updated information to stakeholders using labeling and periodic reports such as Periodic Benefit Risk Evaluation Report (PBRER).
Who should apply
Employees of regulatory authority with working experience in pharmaceutical field over 3 years including at least 1 year in pharmacovigilance.
- The training will be provided in English (with consecutive translation in some sessions).
- All participants are expected to actively participate in all of the sessions.
- A participant from each economy should present some information about pharmacovigilance system in his/her economy to other participants.
Date
February 4 - 7, 2019
Program (subject to change)
See attached the draft program
Registration (Registration is closed.)
Number of participants: approx. 30
Close of registration: November 16, 2018
- Registration request should be made by filling in all the necessary items including signature by Head of Organization on the application form (attached). Please send the form in both the Word file (without signature) and the PDF file (with signature) by e-mail to the e-mail address shown in “Contact Us” below.
- Early registration is recommended. Also, please be sure to allow enough time to obtain the visa for entry to Japan.
- If the number of applications exceeds the capacity, the number of participants from each country may be limited.
- Confirmation of the registration and additional information will be sent to the approved participants after the close of registration.
Fee
There is no registration fee for this seminar.
Information on travel and hotel reservation assistance will be announced to the approved participants.
Location
Pharmaceuticals and Medical Devices Agency (PMDA):
Shin-kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku,Tokyo 100-0013, Japan
Access
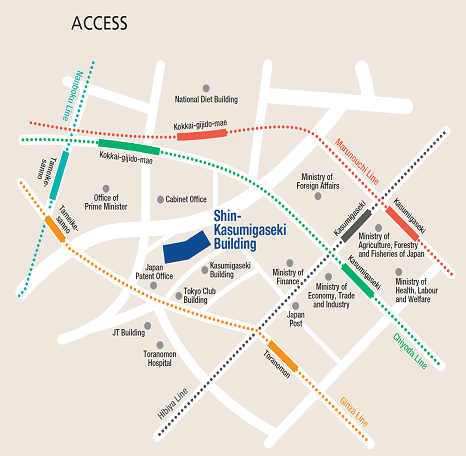
Please use public transportation. Nearest Tokyo Metro Subway Stations:
- 5 minutes' walk from Exit 11 of Toranomon Station on the Ginza Line
- 8 minutes' walk from Exit 3 of Kokkai-gijido-mae Station, on the Marunouchi Line, Chiyoda Line
- 8 minutes' walk from Exit A13 of Kasumigaseki Station, on the Marunouchi Line, Chiyoda Line, Hibiya Line
- 10 minutes' walk from Exit 8 of Tameike-sanno Station, on the Ginza Line, Namboku Line
Contact Us
For more information, please contact:
Secretariat, PMDA Asia Training Center
E-mail: PMDA-ATC●pmda.go.jp
Office of International Cooperation
Pharmaceuticals and Medical Devices Agency
Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan
(Note: For the purpose of security, @ in the e-mail addresses are replaced with ●. Please replace ● with @ when you send an e-mail.)
