
Click here to see the Promotion Flyer [846KB]
Introduction
The Pharmaceuticals and Medical Devices Agency (PMDA) is pleased to announce the “PMDA-ATC & U.S. FDA Pediatric Review Seminar 2026,” coordinated for overseas regulatory authority officials engaged in reviews of pediatric new drug applications. This seminar will be held IN PERSON at the PMDA Office in Tokyo from June 23 to 25, 2026.
The seminar will cover topics related to current pediatric guidelines and practices. The lectures and Q&A sessions are delivered by PMDA and international regulators. Participants will take up case studies, discussing in small groups.
The aim of the seminar is to provide participants with opportunities to take a fresh look at their own regulatory systems and to gather additional information that may enhance the review of pediatric drug development programs within their own regulatory frameworks.
We welcome applications from many regulatory authorities. Please note that the seminar is closed to industry and non-regulatory authorities.
Key Seminar Objectives
- To develop a good understanding of the scientific, ethical and regulatory considerations for evaluating drug products developed for the pediatric population
- To learn the global standard guidelines related to the review of drug products being developed for the pediatric population
- To update knowledge of current practices in the review of drug products being developed for the pediatric population
- To learn modeling and simulation for pediatric drug development
Who should apply
Regulators ONLY (intermediate level)
- Employees of regulatory authorities with working experience of pharmaceuticals review, particularly in the pediatric field for over 5 years
- English will be used in the lectures and discussions during the seminar. Participants must be proficient in English.
Dates / Time
- June 23 (Tuesday), 2026 : 10:00 to 18:00 JST (UTC+9)
- June 24 (Wednesday), 2026 : 9:30 to 16:40 JST (UTC+9)
- June 25 (Thursday), 2026 : 9:30 to 16:30 JST (UTC+9)
Program (subject to change)
See the attached program [124KB]
Applications (Registration Closed)
Number of participants: approx. 30
Registrations close: April 14, 2026
To apply for the seminar, please make sure to complete all necessary items on the application form, including the form with the signature by the applicant's head of the organization, before the closing date. Click the link to register: https://atcform.pmda.go.jp/servlet/front?id=107&p=1&m=1
- Selection of participants will be at the discretion of PMDA.
- Confirmation of registration and additional information will be sent to the invited participants after the application closing date.
Others
- Materials will be provided electronically. Participants are asked to bring their own personal computers to access these materials as needed.
Fee
There is no registration fee for this seminar.
- Information on travel and hotel reservation assistance will be announced to the approved participants.
- International travel insurance is not covered by PMDA; PMDA strongly recommends participants to purchase a plan before departure.
Seminar Venue
Pharmaceuticals and Medical Devices Agency (PMDA)
Shin-kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan
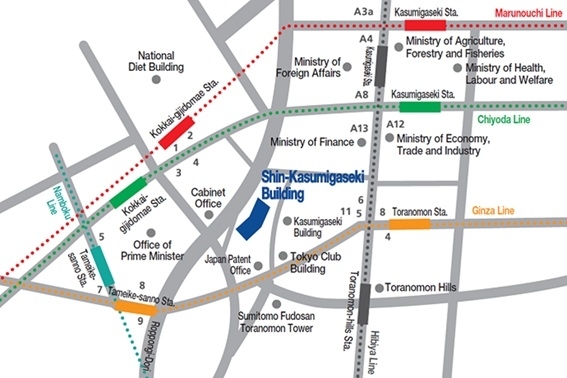
Please use public transportation. Nearest Tokyo Metro Subway Stations:
- Toranomon - Ginza Line (G07) 5 min from Exit 11
- Kokkai-gijido-mae - Marunouchi Line, Chiyoda Line (M14, C07) 8 min from Exit 3
- Kasumigaseki - Marunouchi Line, Chiyoda Line, Hibiya Line (M15, C08, H07) 8 min from Exit A13
- Tameike-sanno - Ginza Line, Namboku Line (G06, N06) 10 min from Exit 8
Contact Us
For more information, please contact:
Secretariat, PMDA Asia Training Center
E-mail: PMDA-ATC[at]pmda.go.jp
Asia Training Center for Pharmaceuticals and Medical Devices Regulatory Affairs
Pharmaceuticals and Medical Devices Agency
Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan
(Note: For security, e-mail address' @ are replaced with [at]. Please replace [at] with @ when sending e-mails to us.)
